Sponsored by
By Jennifer Rood, PhD | Professor and Associate Executive Director for Cores and Resources
With the COVID-19 pandemic, many people are calling for widespread testing. But testing of what and for what?
At first glance, it seems easy enough: Test everyone to see if they have a current infection or if they have been infected in the past and are immune to future infection. However, there are different types of tests, each with its own strengths and weaknesses.
To delve a little bit deeper, let’s break down the tests into two categories: viral (antigen) testing and antibody testing.
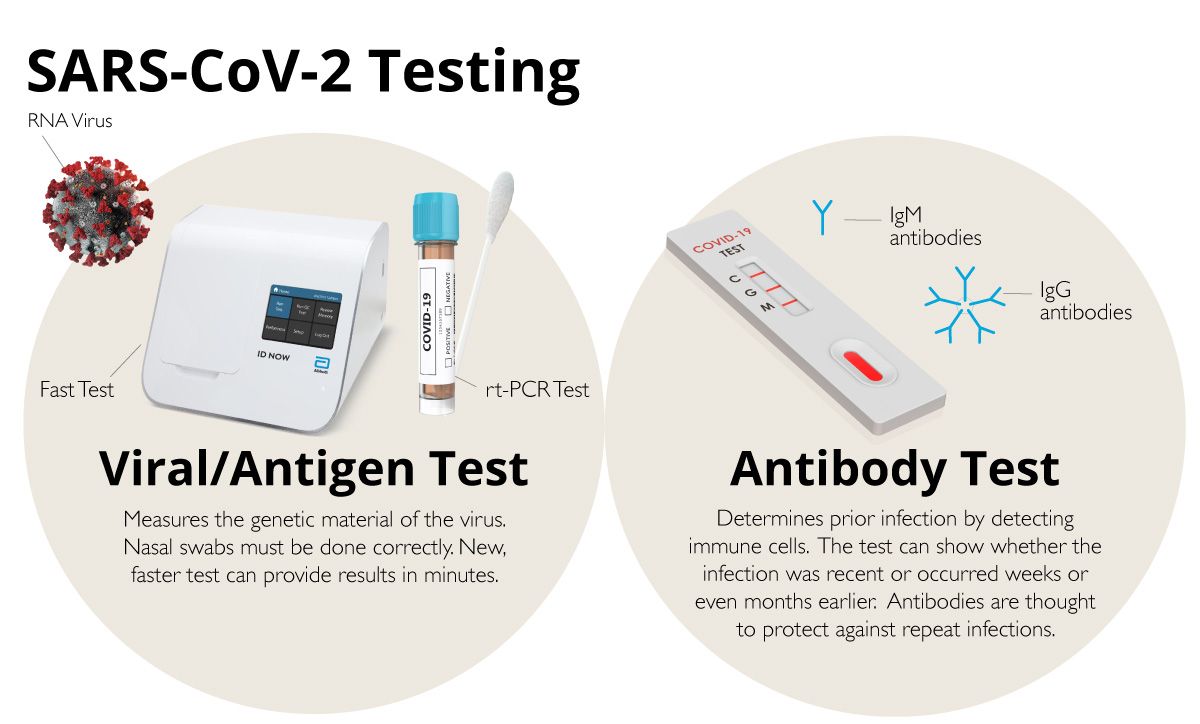
COVID-19 is caused by SARS-CoV-2, which is an RNA virus. When someone has an active infection, an antigen test can measure the genetic material of the virus within his/her body. The virus is primarily found in a person’s airway, so a long swab is inserted into the nose to collect fluid and cells that may contain the viral particles.
Clinical laboratory scientists can extract the genetic material from the swab to look for the presence of the virus using a technique termed rt-PCR. If the sample is not collected properly or if the virus is not present in detectable quantities, then a false negative result can occur.
Inaccurate test results have serious implications for the public. In addition to the rt-PCR tests, newer tests that can be done at the bedside are being released on the market. These tests offer rapid results, but again, a word of caution: Not all of these tests have been reviewed and validated by the FDA, so there is concern about inaccurate results.
A second type of test is called an antibody test. This test is used to determine if the virus has been present in the past. Antibodies are immune cells that are produced in the body after it detects a specific pathogen; in this case, SARS-CoV-2.
Unlike the antigen test, the antibody test generally uses blood samples. It takes several days for antibodies to appear in large enough numbers for measurement. To further complicate the testing paradigm, there are two types of antibodies that can be measured: IgM and IgG. IgM molecules are the initial type of immune cell produced when the body becomes infected. On average, these antibodies are present for five to seven days.
The next immune cells to be produced are IgG antibodies, which can last for months or longer. There are antibody tests that can detect IgG and IgM so one can determine if the infection was recent or further in the past. Again, many of the tests that are being used have not been cleared by the FDA.
Early data show that people who have recovered from COVID-19 and have antibodies are likely to be protected against future infection, but it’s unclear if everyone who has recovered will have immunity or how long that immunity will last.
With each passing day, new antigen and antibody tests are being released, but the demand still far outweighs the supply. In an ideal scenario, the two types of tests—antigen and antibody—should be used to complement one another to check for current and past infection. As more is learned about the virus, the tests will become more reliable and can be used to track and contain the spread of the disease.
Links for more information
Testing for COVID-19
Centers for Disease Control & Prevention
FAQs on Diagnostic Testing for SARS-CoV-2
U.S. Food & Drug Administration
Click here to learn more about Pennington’s role in understanding the critical link between obesity and COVID-19. To support Pennington Biomedical’s important work, please click here.
 GET DAILY REPORT FREE
GET DAILY REPORT FREE