Sponsored by
By Claude Bouchard, PhD
LSU Boyd Professor
Pennington Biomedical Research Center
A large number of people with COVID-19 are asymptomatic. Others recover quickly while many suffer severe symptoms including profound respiratory distress. A significant number die from the infection. What are the driving causes of this variability?
Each one of us carries about 5 million DNA variants in the genome of our cells. It is a small number compared to the approximately 3 billion pairs of DNA bases present in the human genome. However, these DNA variants influence biology (and behavior) because they often change the chemical properties of proteins they code for, alter the level of expression of genes they relate to or modify other molecular events in cells. These DNA variants are also the reason there are genetic predispositions to obesity, hypertension, type 2 diabetes, cardiovascular disease, dementia and infectious diseases.
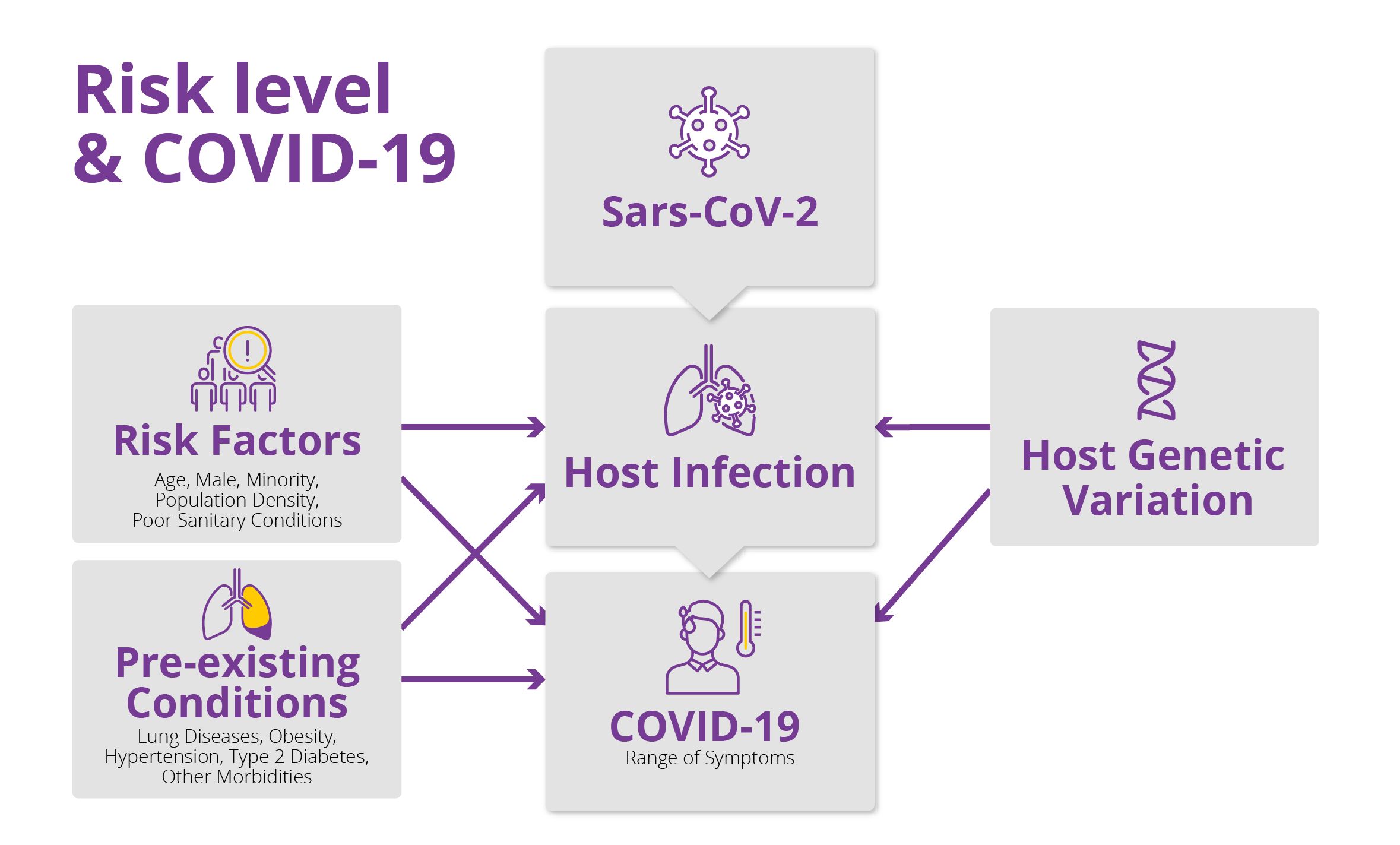
While we are still in the very early stages of investigating COVID-19, research has already shown that older people, adults with pre-existing conditions such as obesity, hypertension type 2 diabetes and other health issues, men, and minorities are more vulnerable than the rest of the population. Now several reports suggest that the individual genetic profile of an individual (the host) can play a role in meeting the challenges posed by the SARS-CoV-2 virus, which causes COVID-19. Scientists are seeking the answers to three critical questions about an individuals’ genetic profile:
• Does it put them at higher or lower risk of infection?
• Does it influence the magnitude of the immune response to the virus?
• Does it affect the severity of the symptoms and increase risk of death?
These questions will keep scientists busy for years but the early evidence strongly suggests that the genetic profile of the host is of great importance.
Host genetics and severity of symptoms. One study from the United Kingdom suggests that identical twins are substantially more alike in symptoms such as fever, loss of taste and smell than fraternal twins. This suggests that there is a genetic contribution to the severity of the symptoms experienced when infected. An issue of great interest is whether the genetic determinants of obesity, hypertension, diabetes and other morbidities known to increase the vulnerability to the viral infection also contribute to the physiological response to the infection itself. This is a topic of great importance that needs to be addressed in depth.
Host genetics and the risk of infection. Prior studies have shown that several DNA variants influence a person’s susceptibility to infection. For example, some people’s genes slow or help fight off HIV, malaria, hepatitis B and other infections. It has been suggested that people with blood type O or with one of several subtypes of tissue antigens (within the HLA system) are more prone to the coronavirus infection compared to those with other HLA genetic characteristics. HLA genes provide the instructions to produce proteins that are present on the cell surface of almost all tissues. These proteins bind to intruders, like the coronavirus, and trigger the host immune response. Some people’s HLA defenses appear more effective against the virus while others’ are less so.
One promising area of investigation is the ACE2 gene, which produces a protein that appears to be the major receptor of SARS-CoV-2. A person’s genetics may determine how accessible the proteins encoded by the ACE2 gene are to the virus. The virus invades the cell by using proteins, known as spike proteins, that are located in the outer covering of the cell. The virus can’t dock to a host cell unless it has receptors that fit the spikes. Your DNA helps determine how many ACE2 receptors there are on your cells, and possibly your risk of infection.
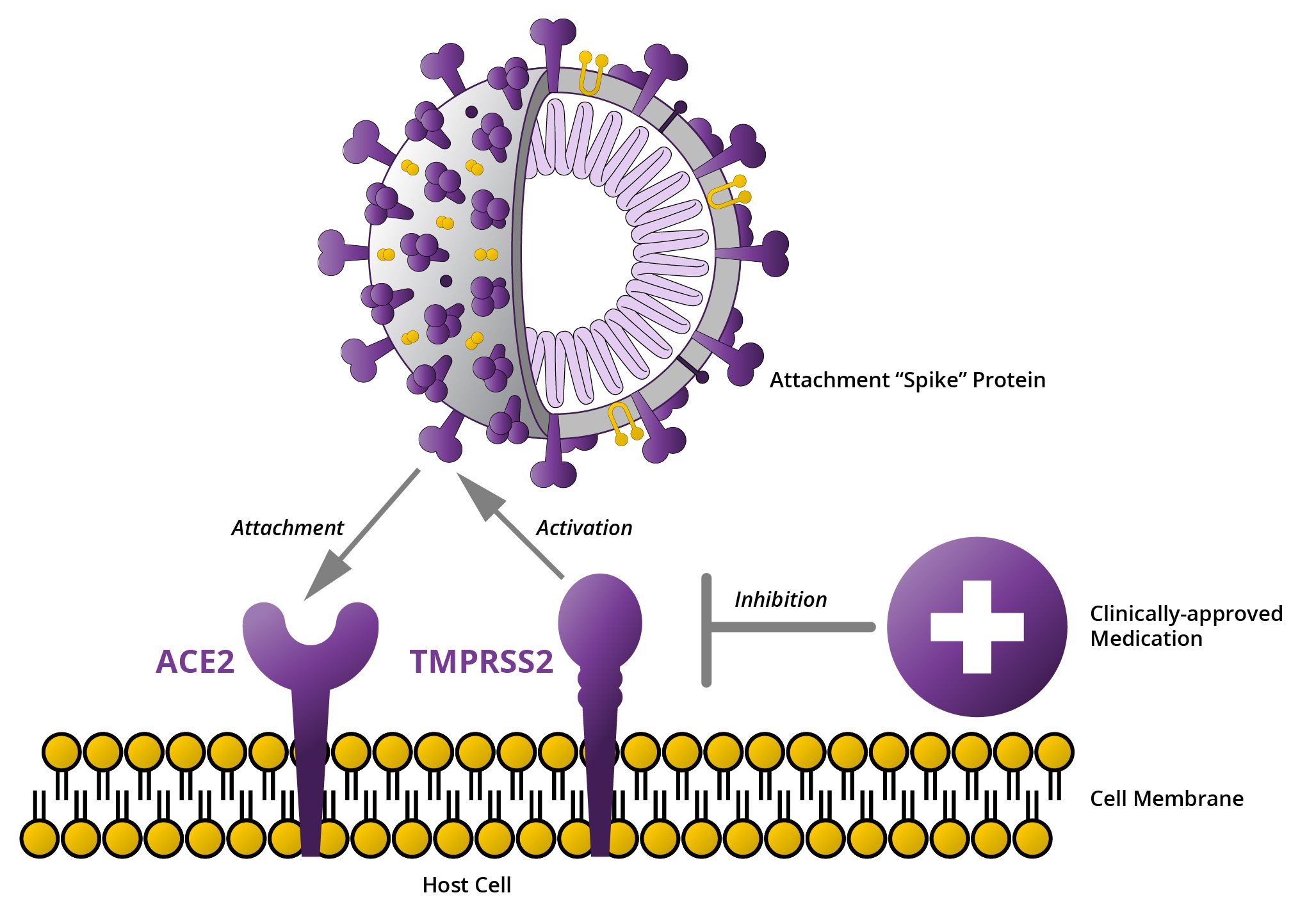
Less than six months have elapsed since the SARS-CoV-2 coronavirus was identified and its infection potential recognized. Remarkable advances have occurred in a very short period of time, but a lot more needs to be done. There is hope on the genetic front that much will be learned in the coming months.
For instance, there are more than 100 ongoing projects worldwide exploring the host genomic signatures of infection risk and the range of symptoms experienced once infected; the effort is being supported by a coordinating team based at the University of Helsinki. In-depth genetic studies of outliers such as young people who experience very severe symptoms, older people who experience no or minor symptoms, infected people with obesity and/or hypertension and/or diabetes who are experiencing no symptoms once infected are also of great interest.
In summary, because it is so early in the research agenda, one cannot use genetic information to guide prevention and treatment as of yet. In the meantime, we should assume that we are not all equally at risk because we are genetically different.
Click here to learn more about Pennington’s role in understanding the critical link between obesity and Covid-19. To support Pennington Biomedical’s important work, please click here.
 GET DAILY REPORT FREE
GET DAILY REPORT FREE